The Food and Drug Administration has approved the powerful narcotic painkiller OxyContin for kids as young as eleven. While doctors who treat young cancer patients hailed the acceptance, others expressed concern that prescribing OxyContin to kids could put them at risk for dependence.
OxyContin, an extended-release version of the painkiller oxycodone, has gained notoriety recently due to its regular misuse. The pills are crushed by people addicted to painkillers so they can be snorted or injected, producing a high that is powerful.
In 2010, Purdue Pharma reformulated OxyContin to make it more challenging to abuse.
The FDA notes that children generally have many fewer choices for pain relief than grownups. Because of that difficulty, the FDA asked Purdue to perform studies to see whether the drug may be utilized safely in kids ages 11 to 16 with pain caused by cancer, trauma or major surgery, said Sharon Hertz, a physician with all the FDA’s Center for Drug Evaluation and Research, in an interview on the agency’s website.
The FDA approved OxyContin for children this age who want “daily, round-the-clock, long-term” pain relief for which there is no option, Hertz said. Doctors should only prescribe OxyContin in children that have already been treated with opiate painkillers and who can stand at least 20 milligrams a day of oxycodone.
Other than OxyContin, the only other long-acting painkiller approved for kids is Duragesic, also known as fentanyl, Hertz said.
“Kids aren’t treated with opioids very commonly and generally it’s just for a small time period with close supervision by health care professionals,” Hertz said.
Doctors who treat pediatric cancer patients hailed the approval as ways to relieve children’s suffering. Kids at the end of life are not at risk of dependence.
Long acting medications prevent breakthrough pain, so that children can feel comfortable and “focus their energy on truly being a kid instead of fighting their pain,” Baker said.
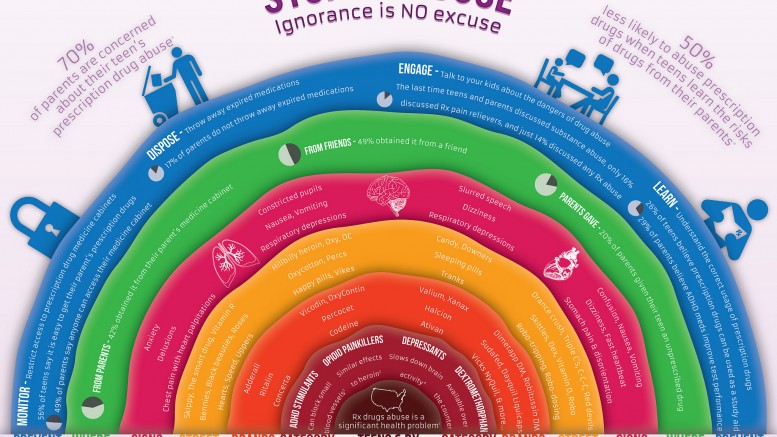
But prescribing OxyContin to children with short term medical needs could be put them at risk for developing an addiction that haunts them long after they leave the hospital, said Andrew Kolodny, manager of Physicians for Responsible Opioid Prescribing. Teens are at higher danger of dependence than adults because the brain doesn’t mature until about age 25. Studies show that about one in 25 high school seniors has abused OxyContin, said Scott Hadland, a specialist in substance abuse treatment and adolescent medicine at Boston Children’s Hospital and Harvard Medical School.
[wp_ad_camp_3]
“Among youths who are prescribed OxyContin, a modest but significant amount are likely to become addicted,” Hadland said.
The amount of prescription painkillers sold in the USA has quadrupled since 1999, based on the Centers for Disease Control and Prevention. More than 44,000 Americans die of drug overdoses each year. Some people who become addicted to prescription painkillers change to using heroin, which has become easier and more affordable to access than OxyContin.
Kolodny said it is concerning the FDA approved OxyContin for children without appointing an advisory panel to talk about the dangers and benefits, a process traditionally used when the bureau confronts a choice that was contentious.
Hadland said physicians should take special precautions when dispensing painkillers, including prescribing small amounts, in order that people do not end up with extra pills that they do not desire. Before prescribing OxyContin doctors should screen patients for alcohol and drug misuse, Hadland said. And doctors should assess their state’s prescription drug monitoring program, which allows them to see if painkillers have been received by patients from some other physicians.
Parents should be responsible for giving kids the drug, rather than enabling teens to administer their own painkillers, Hadland said. Painkillers should be kept by parents.
REFERENCES
1. Food And Drug Administration. CDER Conversation: Pediatric pain management options. (Accessed October 2015)
2. Catherine Saint Louis. F.D.A. Approval of OxyContin Use for Children Continues to Draw Scrutiny. New York Times, October 8, 2015.
3. Food and Drug Administration. Letter to Purdue Pharma. (Accessed October 2015)